The atomic volume is not the volume that an atom occupies, but refers to the volume occupied by a fixed quantity of a given number of atoms of a chemical element.
In the text Chemical element, it was shown that an element corresponds to a set of atoms that have the same atomic number, that is, the same amount of protons in the nucleus. The quantity established to calculate the atomic volume is the value of Avogadro's constant, which is equal to 6.02. 1023 atoms, which corresponds to 1 mole of the element.
So we can say that the atomic volume is the volume that 1 mole of atoms (or 6.02). 1023 atoms) of an element occupies.
This quantity can be calculated if we know the density of the solid-state element and the mass of 1 mole of atoms of that element. For example, consider that the density of sodium (Na) is equal to 0.97 g/cm3 and that 6.02. 1023 sodium atoms have a mass equal to 23.0 g. We can then, from the formula of density, arrive at the atomic volume of sodium. See how:
density = pasta
volume
atomic volume = mass of 6.02. 1023 element atoms
solid state element density
Atomic volume of sodium = 23.0 g
0.97 g/cm3
Atomic volume of sodium = 23.71 cm3
The atomic volume value involves not only the size of the atoms, but also the distance between them. Therefore, since it also involves empty spaces, we can not take the atomic volume value and divide by the number of atoms (6.02. 1023) to find the volume each atom occupies.
It is important to study the atomic volume because it is a periodic property, that is, it varies according to the atomic number of the element. Look at the image below. The atomic volume of the elements grows from top to bottom, which means that when we analyze the elements belonging to the same family of the Periodic Table, as the atomic number grows, the atomic volume also grows. This is because, in this sense, the periods increase, which means that the electronic layers of the atoms increase and, consequently, the volume occupied by them also increases. This is the same sense of growth as atomic ray.
On the other hand, horizontally, we see that the atomic volume increases from the center to the edges. When we start from the left and go right to the middle of the Periodic Table, the atomic volume decreases because the period is the same, that is, the elements of the same line have the same amount of electronic layers, but the atomic number will increasing. This means that the amount of electrons increases in this direction, which results in a greater force of attraction of electrons to the nucleus of the atom, decreasing its atomic radius and, consequently, the volume atomic. But from the center to the right, this does not continue to happen, because, in the case of elements located in this region, especially in the case of non-metals, the spacing between their atoms is relatively large, which affects the atomic volume, increasing it.
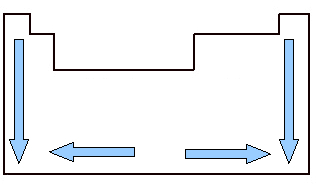
Atomic volume variation in the Periodic Table across families and periods
By Jennifer Fogaça
Graduated in Chemistry
Source: Brazil School - https://brasilescola.uol.com.br/quimica/volume-atomico.htm
