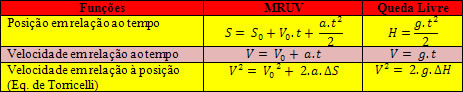
The Third Law of Thermodynamics deals with the behavior of matter with an entropy approaching zero.
According to this law, whenever a system is in thermodynamic equilibrium, its entropy approaches zero.
The second law of thermodynamics relates to entropy. Subsequently, the third law appears as a attempt to establish an absolute reference point that determines the entropy.
Walther Nernst (1864-1941) was the physicist who dealt with the principles that formed the basis for the third law of thermodynamics.
According to Nernst, entropy would tend to have a minimum value if the temperature of a pure substance were equal to or approaching absolute zero.
For that, Nernst proposed the formula below, which shows that the variation of entropy (ΔS) and temperature (T) tend to minimum values, that is, 0:
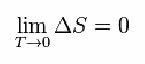
But what is Entropy?
Entropy it's the way molecules organize themselves in the system. This organization translates into disorder, not in the sense of confusion, but in the sense of movement and agitation of molecules.
The more molecules can move, the more disorganized they are, the more entropy they have.
Initially, Nernst suggested that the entropy he proposed would be possible only on perfect crystals.
Finally, he concluded that the temperature equal to absolute zero did not exist, which makes the third law a controversial law.
So, for many physicists, this is not a law but a rule.
After so many years (since 1912), scientists try to obtain this temperature or temperatures that come closer and closer to absolute zero. Thus, they discovered that it is only possible in gases, discarding any substance in solid or liquid state.
Read too:
- Zero Law of Thermodynamics
- First Law of Thermodynamics
- Second Law of Thermodynamics
- Exercises on Thermodynamics