Several chemical reactions in our daily lives occur with loss and gain of energy in the form of heat, as the formation and breaking of bonds involve interaction of energy with matter. In addition to reactions, this also occurs in physical state changes.
exothermic processes
A common reaction of combustion is the burning of wood. It is known that this process occurs with the release of energy in the form of heat as well as light. Thus, this reaction is called exothermic (the prefix “exo” means “outside”), as heat is released.
One exothermic process which is not a reaction, but rather a change in the state of aggregation, is the formation of snow. Liquid water solidifies, forming snow when heat is released. In this process, more specifically, 7.3 kJ of heat is released.
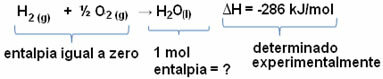
in these exothermic processes, the variation of enthalpy (ΔH), that is, the amount of heat released will always be negative (∆H< 0), because the enthalpy variation is measured by decreasing the enthalpy of the products by the enthalpy of the reagents:
ΔH = Hproducts- Hreagents
Do not stop now... There's more after the advertising ;)
or
ΔH = HFinal- Hinitial
As there was energy released, the enthalpy of the products will be lower and, therefore, the enthalpy variation will be negative. Other examples of exothermic reactions and phenomena are listed below:

endothermic processes
Reactions or changes in physical state that absorb heat are called endothermic processes. An example of endothermic reaction (the prefix “endo” means “inside”) is the cooking of food, in which energy is needed. On the other hand, a physical state transformation that is endothermic is the evaporation of liquid water, which, in order to occur, needs to absorb 44 kJ.
In this case, as the enthalpy of the products or the final state increases, as energy is absorbed, the enthalpy variation is always positive (ΔH > 0). Note other examples of endothermic processes:

In short:
US exothermic processes, the system loses heat and the environment is heated;
US endothermic processes, the system gains heat and the environment cools down.
By Jennifer Fogaça
Graduated in Chemistry
Would you like to reference this text in a school or academic work? Look:
FOGAÇA, Jennifer Rocha Vargas. "Endothermic and exothermic processes"; Brazil School. Available in: https://brasilescola.uol.com.br/quimica/processos-endotermicos-exotermicos.htm. Accessed on June 27, 2021.
Chemistry

What will hell be like according to Boyle's Law, Hell is endothermic or exothermic, processes endothermics and exothermics, heat absorption, heat release, pressure product, constant temperature, Boyle's Law.